Cancer Stem Cells
The Cells Oncologists Don't want to talk about
Cancer stem cells (CSCs) are a subset of cancer cells that exhibit characteristics similar to those of normal stem cells, including self-renewal and the ability to differentiate into various cell types within a tumor. These cells are believed to drive tumor initiation, progression, metastasis, and recurrence due to their ability to evade conventional treatments and regenerate tumors. The cell of origin of CSCs is unknown and widely debated. It is postulated that tissue-resident stem cells (TRSCs) transform into CSCs through a combination of genetic mutations, epigenetic changes, and microenvironmental interactions.(1-5)
Biochemical Pathways Involved in CSCs
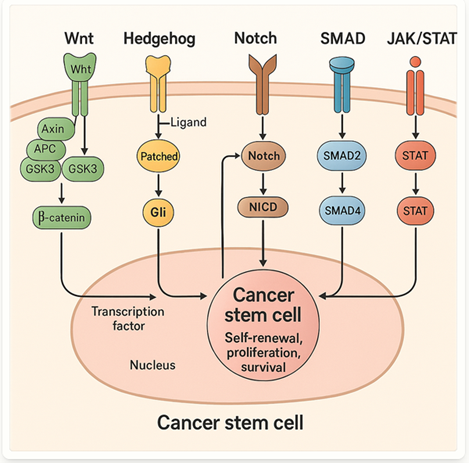
Several key signaling pathways (see Figure 1) are involved in the regulation and maintenance of CSCs, including:
Wnt/β-catenin pathway: Crucial for CSC self-renewal and differentiation
Hedgehog pathway: Involved in CSC proliferation, self-renewal, and tumorigenicity
Notch pathway: Regulates CSC replication, survival, and differentiation
TGF-β/SMAD pathway: Promotes CSC self-renewal, migration, and invasion
JAK/STAT pathway: Contributes to CSC growth and sustained inflammation in the tumor microenvironment
Figure 1. Cancer stem cell pathways
CSC Metabolic Flexibility
CSCs exhibit metabolic flexibility, utilizing both glycolysis and oxidative phosphorylation (OXPHOS), depending on tumor type, microenvironment, and genetic factors. While aerobic glycolysis (the Warburg effect) is common in many cancers, CSCs often retain functional mitochondrial OXPHOS and can switch between these pathways to meet energy demands and survive under stress.
Effects of Chemotherapy and Radiation Therapy on CSCs
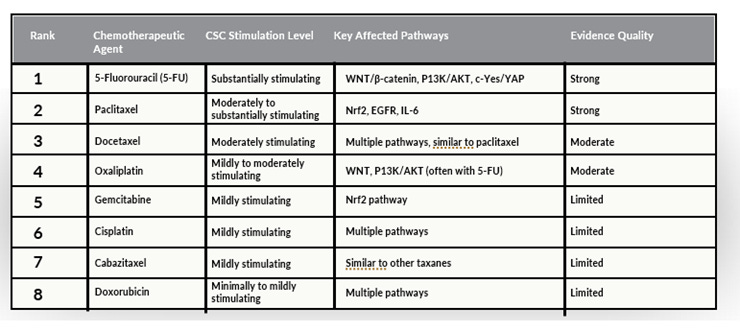
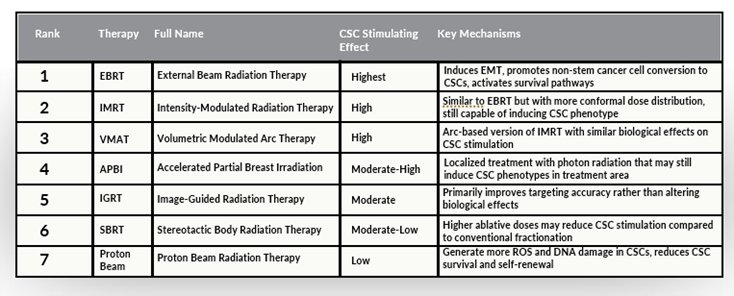
Conventional chemotherapy and radiation therapy primarily target rapidly dividing cells, which form the bulk of a tumor. However, because CSCs constitute only a small fraction of the tumor, their quiescent nature and enhanced resistance mechanisms often allow them to evade treatment. This can lead to tumor recurrence, as CSCs are capable of regenerating the tumor. More concerning, some chemotherapy drugs and certain forms of radiation therapy may actually activate CSCs (see Tables 1 and 2).
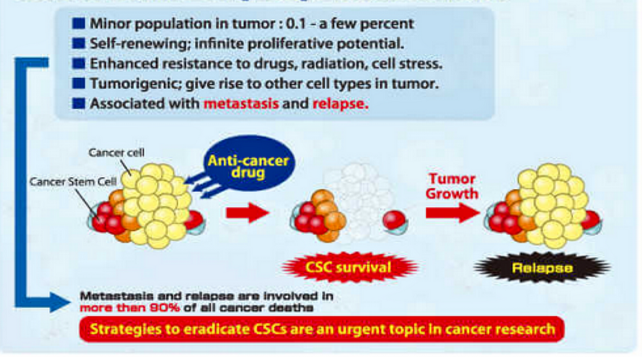
The clinical consequences of CSC resistance—including relapse and metastasis—are illustrated in Figure 2.
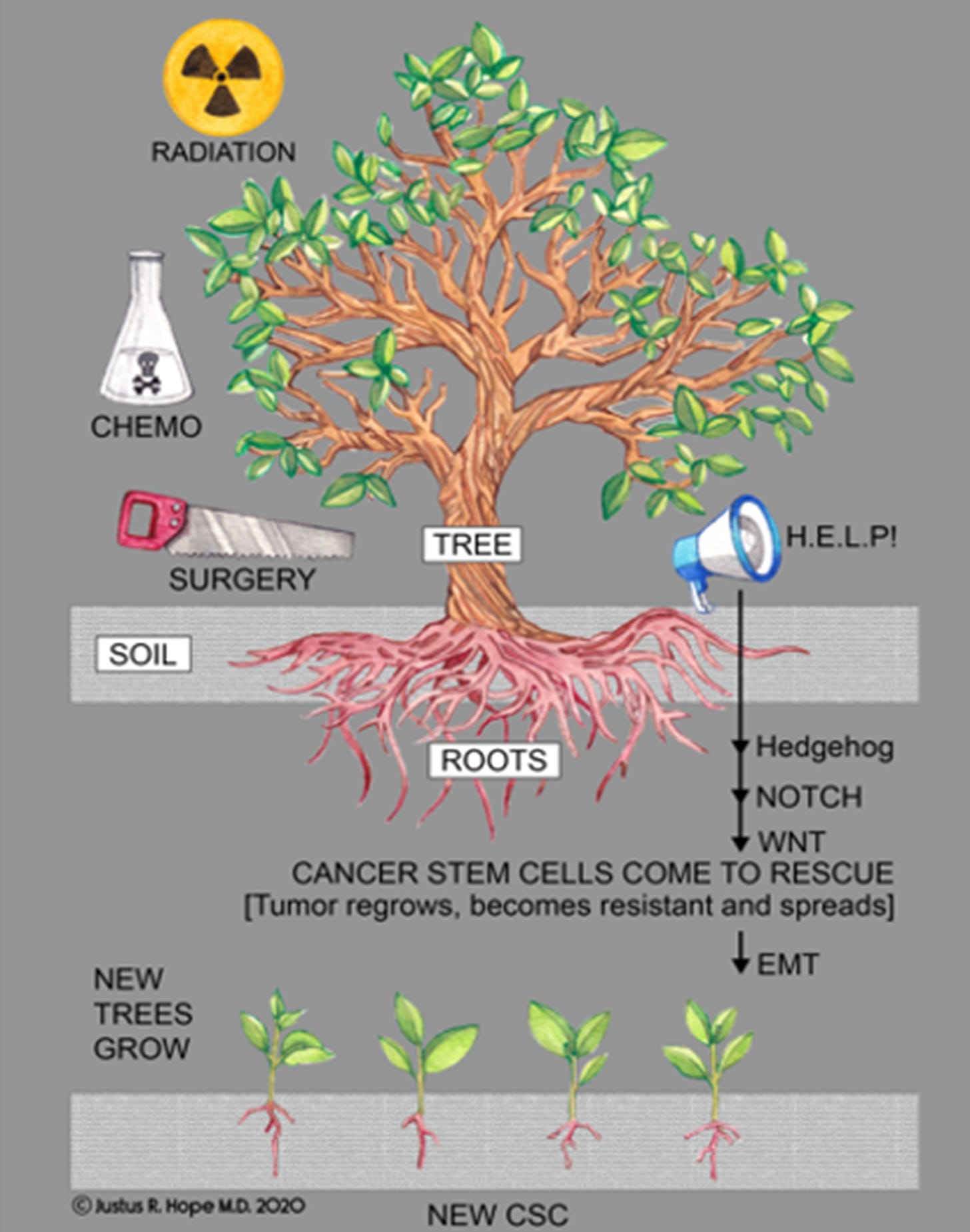
Figure 3 provides a visual analogy of how CSCs (like the roots of a tree) survive chemotherapy, radiation, and surgery, leading to relapse and continued tumor growth.
Figure 2. Characteristics of cancer stem cells (CSCs)
Figure 3. Cancer stem cells: Visual analogy of resistance and regrowth.
Why Are CSCs Resistant to Chemotherapy?
CSCs resist chemotherapy through multiple interconnected biological mechanisms that allow them to survive treatment and drive tumor relapse. These mechanisms include both intrinsic adaptations and interactions with the tumor microenvironment (TME), as outlined below:(6, 7)
1. Intrinsic Resistance Mechanisms
Drug efflux pumps: CSCs overexpress ATP-binding cassette (ABC) transporters such as ABCB1 and ABCG2, which actively pump chemotherapeutic agents out of the cell. This reduces intracellular drug accumulation and contributes to the multidrug resistance (MDR) phenotype—a hallmark of CSCs.
Quiescence (dormancy): CSCs often enter a slow-cycling or nondividing state, allowing them to evade therapies that target rapidly proliferating cells. For example, glioblastoma CSCs survive temozolomide through phenotypic interconversion between dormant and active states.
Enhanced DNA repair: CSCs exhibit upregulation of DNA damage response pathways, including ATM/ATR kinases and CHK1/CHK2 checkpoints. This enables efficient repair of DNA damage caused by chemotherapy or radiation.
Anti-apoptotic signaling: CSCs overexpress anti-apoptotic proteins such as BCL-2 while suppressing pro-apoptotic signals, thereby enabling resistance to chemotherapy-induced cell death.
2. Extrinsic Resistance Mechanisms
Tumor microenvironment (TME) support: The TME—comprising adipocytes, fibroblasts, and immune cells—secretes growth factors (e.g., VEGF, FGF) and cytokines that promote CSC survival and stemness. For example, VEGF-NRP1 signaling in glioblastoma enriches chemoresistant CSCs following bevacizumab treatment.
Hypoxia: CSCs thrive in hypoxic niches, where hypoxia-inducible factors (HIFs) are activated and promote stemness while upregulating drug efflux pumps.
Epithelial-mesenchymal transition (EMT): EMT enhances CSC plasticity, invasiveness, and resistance by upregulating stemness markers (e.g., CD44, ALDH1) and survival pathways like Wnt/β-catenin and Hedgehog. For instance, Wnt activation in breast CSCs increases ABCB1 expression.
3. Signaling Pathways Driving Resistance
CSCs hijack key developmental pathways to maintain chemoresistance:
Hedgehog: Promotes survival in glioblastoma and colorectal CSCs
Wnt/β-catenin: Upregulated in colorectal and breast CSCs; linked to 5-fluorouracil resistance
Hippo-YAP/TAZ: Sustains stemness in breast cancer and cooperates with Wnt signaling
Table 1. Cancer stem cell activation by chemotherapeutic agents
Table 2. CSC activation by radiotherapy
Repurposed Drugs Inhibiting CSCs
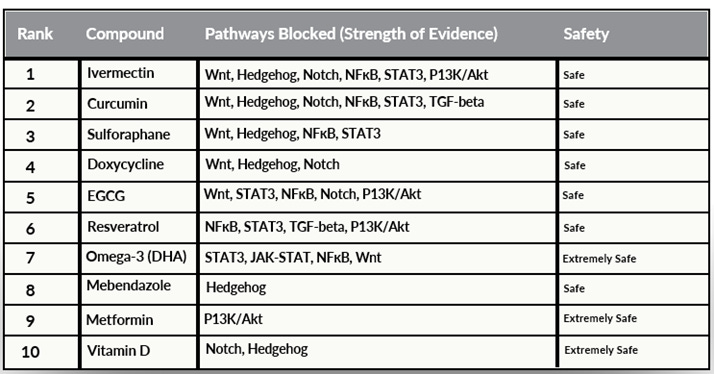
Several repurposed drugs and natural compounds have been found to inhibit CSCs through various signaling and metabolic pathways (see Table 3).(8-21)
Ivermectin: Known for its antiparasitic properties, ivermectin has shown potential to inhibit CSCs by targeting various signaling pathways.
Curcumin: A compound in turmeric with anti-inflammatory and anticancer properties; it inhibits CSCs through pathways such as Wnt/β-catenin.
Mebendazole: An antiparasitic drug explored for its anticancer effects, including targeting CSCs.
EGCG (epigallocatechin gallate): A polyphenol most abundant in green tea; it inhibits CSCs by modulating signaling pathways such as Wnt and Notch.
Doxycycline: An antibiotic studied for its potential to inhibit CSCs by targeting specific pathways.
Metformin: Commonly used to treat diabetes, metformin has been investigated for anticancer effects, including targeting CSCs via metabolic pathways.
Vitamin D: Known for its role in bone health, vitamin D has been studied for its potential to inhibit CSCs by modulating signaling pathways.
Sulforaphane: Found in cruciferous vegetables, sulforaphane has shown potential to inhibit CSCs through various mechanisms.
Resveratrol: A polyphenol found in grapes, resveratrol has anticancer properties and inhibits CSCs by targeting multiple pathways.
These compounds offer promising avenues for developing targeted therapies against CSCs, potentially improving cancer treatment outcomes by reducing recurrence and metastasis. All patients with cancer MUST be treated with an agent active against cancer stem cells.
Table 3. Repurposed agents active against CSCs and pathways blocked
References.
1. Nassar D, Blanpain C. Cancer Stem Cells: Basic Concepts and Therapeutic Implications. Annu. Rev Pathol. 2016;11:47–76.
2. Skvortsova I. Cancer Stem Cells: What Do We Know about Them? Cells. 2021;10(6).
3. Dawood S, Austin L, Cristofanilli M. Cancer stem cells: implications for cancer therapy. Oncology (Williston. Park). 2014;28(12):1101–7, 10.
4. Walcher L, Kistenmacher AK, Suo H, Kitte R, Dluczek S, Strauß A, et al. Cancer Stem Cells-Origins and Biomarkers: Perspectives for Targeted Personalized Therapies. Front Immunol. 2020;11:1280.
5. Butti R, Gunasekaran VP, Kumar TVS, Banerjee P, Kundu GC. Breast cancer stem cells: Biology and therapeutic implications. Int. J Biochem. Cell Biol. 2019;107:38–52.
6. He D, Wu Q, Tian P, Liu Y, Jia Z, Li Z, et al. Chemotherapy awakens dormant cancer cells in lung by inducing neutrophil extracellular traps. Cancer Cell. 2025.
7. D’Alterio C, Scala S, Sozzi G, Roz L, Bertolini G. Paradoxical effects of chemotherapy on tumor relapse and metastasis promotion. Semin Cancer Biol. 2020;60:351–61.
8. Naujokat C, McKee DL. The “Big Five” Phytochemicals Targeting Cancer Stem Cells: Curcumin, EGCG, Sulforaphane, Resveratrol and Genistein. Curr Med Chem. 2021;28(22):4321–42.
9. Joe NS, Godet I, Milki N, Ain NUI, Oza HH, Riggins GJ, et al. Mebendazole prevents distant organ metastases in part by decreasing ITGβ4 expression and cancer stemness. Breast Cancer Res. 2022;24(1):98.
10. Fong D, Christensen CT, Chan MM. Targeting Cancer Stem Cells with Repurposed Drugs to Improve Current Therapies. Recent Pat Anticancer Drug Discov. 2021;16(2):136–60.
11. Srivastava RK, Tang SN, Zhu W, Meeker D, Shankar S. Sulforaphane synergizes with quercetin to inhibit self-renewal capacity of pancreatic cancer stem cells. Front Biosci (Elite Ed). 2011;3(2):515–28.
12. Rodova M, Fu J, Watkins DN, Srivastava RK, Shankar S. Sonic hedgehog signaling inhibition provides opportunities for targeted therapy by sulforaphane in regulating pancreatic cancer stem cell self-renewal. PLoS One. 2012;7(9):e46083.
13. Castro NP, Rangel MC, Merchant AS, MacKinnon G, Cuttitta F, Salomon DS, et al. Sulforaphane Suppresses the Growth of Triple-negative Breast Cancer Stem-like Cells In vitro and In vivo. Cancer Prev Res (Phila). 2019;12(3):147–58.
14. Choi YS, Cho HJ, Jung HJ. Atorvastatin inhibits the proliferation of MKN45-derived gastric cancer stem cells in a mevalonate pathway-independent manner. Korean J Physiol Pharmacol. 2022;26(5):367–75.
15. Bonuccelli G, Sotgia F, Lisanti MP. Matcha green tea (MGT) inhibits the propagation of cancer stem cells (CSCs), by targeting mitochondrial metabolism, glycolysis and multiple cell signalling pathways. Aging (Albany NY). 2018;10(8):1867–83.
16. Shi P, Liu W, Tala, Wang H, Li F, Zhang H, et al. Metformin suppresses triple-negative breast cancer stem cells by targeting KLF5 for degradation. Cell Discov. 2017;3:17010.
17. Li Y, Zhang T. Targeting cancer stem cells by curcumin and clinical applications. Cancer Lett. 2014;346(2):197–205.
18. Scatena C, Roncella M, Di PA, Aretini P, Menicagli M, Fanelli G, et al. Doxycycline, an Inhibitor of Mitochondrial Biogenesis, Effectively Reduces Cancer Stem Cells (CSCs) in Early Breast Cancer Patients: A Clinical Pilot Study. Front Oncol. 2018;8:452.
19. Zhang L, Xu L, Zhang F, Vlashi E. Doxycycline inhibits the cancer stem cell phenotype and epithelial-to-mesenchymal transition in breast cancer. Cell Cycle. 2017;16(8):737–45.
20. Saini N, Yang X. Metformin as an anti-cancer agent: actions and mechanisms targeting cancer stem cells. Acta Biochim. Biophys. Sin. 2018;50:133–43.
21. Li Y, Zhang T. Targeting cancer stem cells with sulforaphane, a dietary component from broccoli and broccoli sprouts. Future Oncol. 2013;9(8):1097–103.

Twice I tried to talk to my oncologist about CSC’s and both times she did not utter a word back about them. She knew I was right. I had TNBC, Stage 3, Grade 3. I immediately started taking IVM and Fenben, along with several other supplements on the list. After just 2 chemo treatments, the tumor and lymph nodes began melting away. I was finally able to get an MRI last week, after 9 of 12 treatments. Results…”RESOLUTION” of breast mass and all lymph nodes. No more chemo for me. Now some minor surgery to remove the clips and further test the tissue for residual disease. I believe it will all be clean. Thank you Dr. Marik for your work!
Profound information Dr. Marik. So very grateful for the information. Many Blessings