CAUTION: Methylene Blue, Red Light Therapy, and Cancer: A Powerful Combination — But In Which Direction?
First, Do No HARM
Interruption of usual programming.
I’ve been asked the same question over and over again in recent weeks.
At first, I didn’t think much of it.
Then I realized something unsettling:
I was making the same mistake myself.
What follows is critically important.
Because in medicine, there is one rule that comes before all others:
First, do no harm.
Secondly, I want to personally thank those of you with paid subscriptions. Your support allows me to continue doing the work I believe in—and, quite simply, to sustain it.
If you’re currently a free subscriber, I would ask you to consider upgrading. Your support directly makes this work possible.
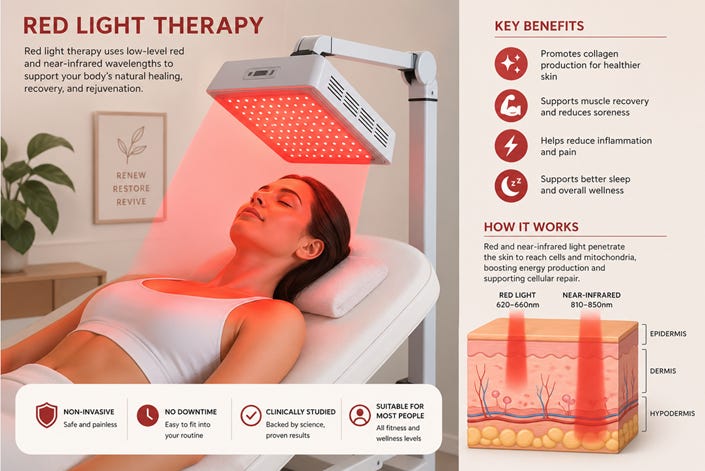
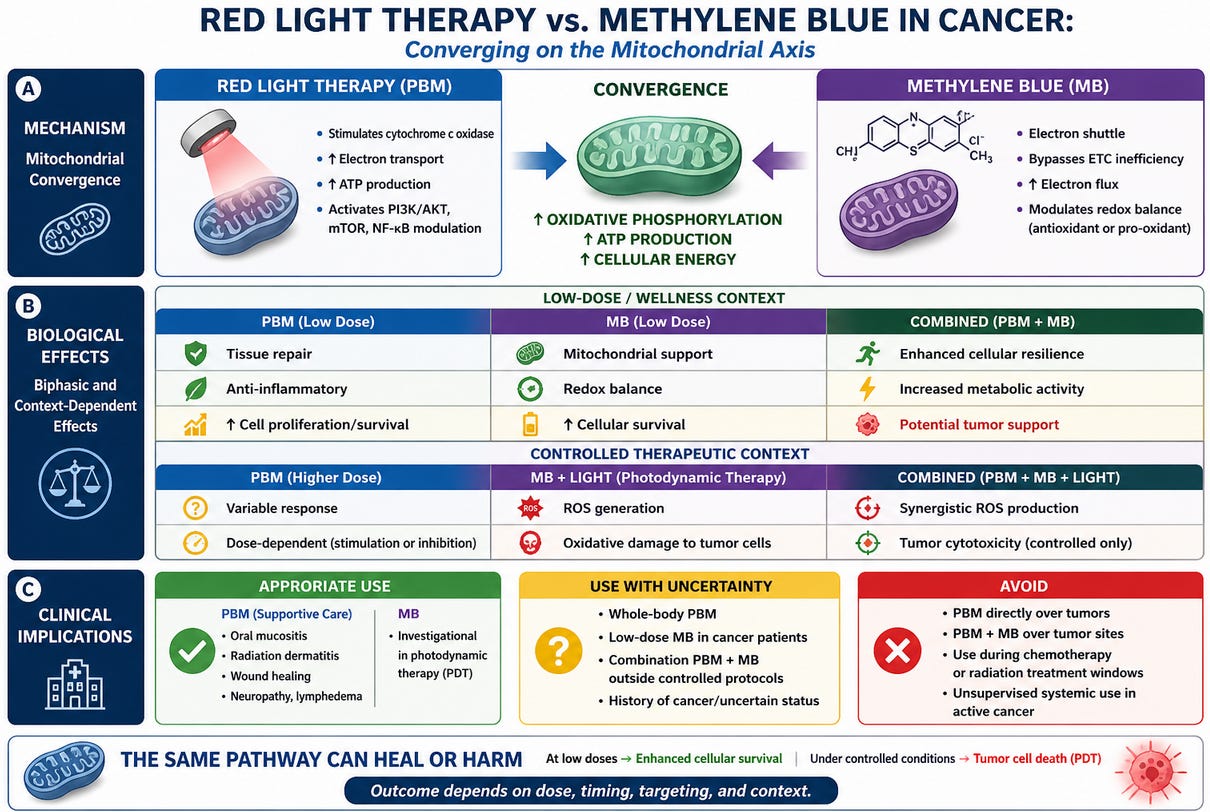
Red light therapy—more precisely photobiomodulation (PBM)—has become enormously popular. Add methylene blue, and you now have a “next-level” biohacking intervention promoted for energy, cognition, and cellular health.
But in oncology, this combination deserves far more caution than enthusiasm.
Because what you are doing—whether you realize it or not—is directly manipulating mitochondrial function, the very engine that determines whether a cancer cell lives or dies.
This combination may salvage cancer cells. You may inadvertently be resuscitating the cancer cell from energy failure and promoting cancer cell proliferation .
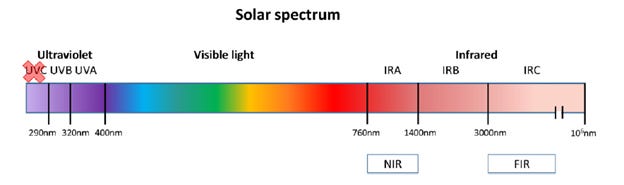
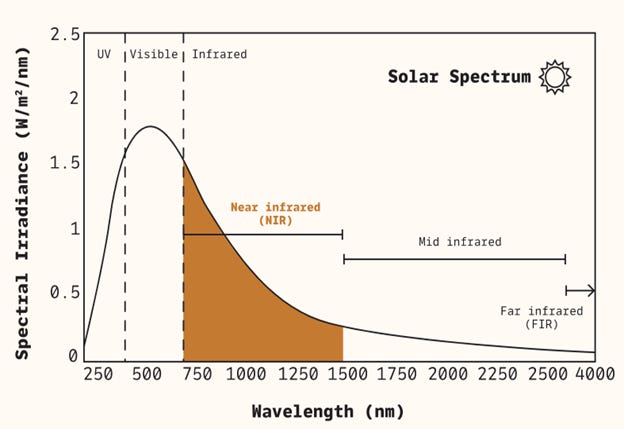
What Is Red Light Therapy?
Photobiomodulation uses specific wavelengths of light to influence cellular function:
Red light: 600–700 nm
Near-infrared light: 700–1000 nm
Its primary biological target is the mitochondrion—specifically cytochrome c oxidase, a key enzyme in cellular respiration.
The downstream effects are well described:
Increased ATP production
Enhanced mitochondrial activity
Reduced oxidative stress (at appropriate doses)
Improved tissue repair and regeneration
Modulation of inflammation
In healthy or damaged tissue, these effects are often beneficial. In cancer, however, the story becomes more complicated.
Where Red Light Therapy Clearly Works
There are several areas in oncology where PBM is not experimental—it is clinically validated and, in some centers, standard supportive care.
1. Oral Mucositis
This is the strongest and most consistent application.
In patients undergoing chemotherapy or radiation—particularly for head and neck cancers—PBM has been shown to:
Reduce severity and duration of mucositis
Decrease pain
Improve oral intake
These benefits are not theoretical; they are reproducible and clinically meaningful.
2. Radiation Dermatitis
PBM can also reduce skin damage from radiation therapy:
Less inflammation
Faster healing
Improved tissue integrity
Again, this is a supportive role—but an important one.
3. Neuropathy and Wound Healing
Emerging evidence suggests benefit in:
Chemotherapy-induced peripheral neuropathy
Surgical wound healing
Possibly lymphedema (though data are less robust)
While not yet definitive, the signal here is promising.
The Central Question: Could PBM Stimulate Cancer Growth?
This is the critical issue—and it cannot be ignored.
The concern arises from basic biology.
PBM works by stimulating mitochondria. In doing so, it:
Increases ATP production
Activates cellular signaling pathways such as PI3K/AKT and mTOR
These are not neutral pathways. They are the very pathways cancer cells exploit to grow, survive, and resist therapy.
This creates a fundamental tension:
In normal tissue: stimulation promotes healing
In cancer cells: the same stimulation could, in theory, promote growth
What Does the Evidence Actually Show?
The data are mixed—and highly dependent on context.
1. Cell Culture Studies
In vitro studies show conflicting results:
Some demonstrate increased tumor cell proliferation and survival
Others show no effect—or even inhibitory effects
The key variable appears to be dose and exposure conditions.
2. Animal Studies
Animal data are similarly inconsistent:
Some studies suggest tumor stimulation at certain doses
Others show no effect—or even immune-mediated anti-tumor effects
3. Human Data
This is where things become more reassuring—but still incomplete.
There is no strong clinical evidence that PBM accelerates cancer progression
However, studies are limited
Long-term safety data are lacking
In other words: absence of evidence is not evidence of absence.
The Clinical Reality
Based on current data, PBM occupies a very specific role:
Clearly safe and beneficial for supportive care
Uncertain when applied directly over tumors
This distinction is crucial.
A Metabolic Perspective: Why Caution Is Warranted
When viewed through a metabolic framework, the potential contradiction becomes even clearer.
Many metabolic cancer strategies aim to:
Reduce mitochondrial efficiency
Increase metabolic stress
Limit ATP availability
PBM does the opposite:
Enhances mitochondrial function
Increases ATP production
Supports cellular energy metabolism
This raises an important concern:
Could PBM counteract therapies designed to metabolically suppress cancer?
This is particularly relevant in tumors that are:
Mitochondria-dependent
Slow-growing but metabolically adaptable
Examples include certain neuroendocrine tumors, where enhancing mitochondrial activity may theoretically provide a growth advantage.
How Should PBM Be Used in Cancer Patients?
A practical, clinically grounded approach is essential.
Appropriate Use
PBM is highly appropriate for:
Oral mucositis
Radiation-induced skin injury
Wound healing
Neuropathy
In these settings, the benefits are clear and the risks are minimal.
Where Caution Is Needed
It is prudent to avoid direct PBM exposure over known tumor sites, particularly:
Primary tumors
Active metastatic lesions
This is not because harm has been definitively proven—but because the biological rationale for caution is strong.
Timing Matters
Another important consideration is timing:
Avoid PBM immediately around tumor-directed therapies
There is a theoretical risk that PBM could protect tumor cells from oxidative or metabolic stress
Using PBM away from treatment windows is a more cautious strategy.
Whole-Body Use
Whole-body PBM remains poorly studied in cancer patients.
Given the systemic increase in mitochondrial activity, its use in active cancer should be approached with restraint until better data are available.
Practical Clinical Framework
Use PBM for:
Mucositis
Radiation skin injury
Pain and neuropathy
Tissue healing
Avoid:
Direct irradiation over tumors
Indiscriminate whole-body use in active cancer
Use immediately around chemotherapy or radiation
Bottom Line
Red light therapy is neither a miracle cure nor a dangerous myth—it is a tool. And like any tool, its value depends on how it is used.
It has strong evidence for symptom management
It plays an important role in supportive oncology care
It is not an anticancer therapy
Its effects on tumor biology remain uncertain
The central concern is biologically plausible:
by enhancing mitochondrial function, PBM may also enhance the very systems cancer cells rely on to survive.
Until more definitive data are available, the most rational approach is one of targeted use and disciplined restraint.
Red light Therapy and Methylene blue.
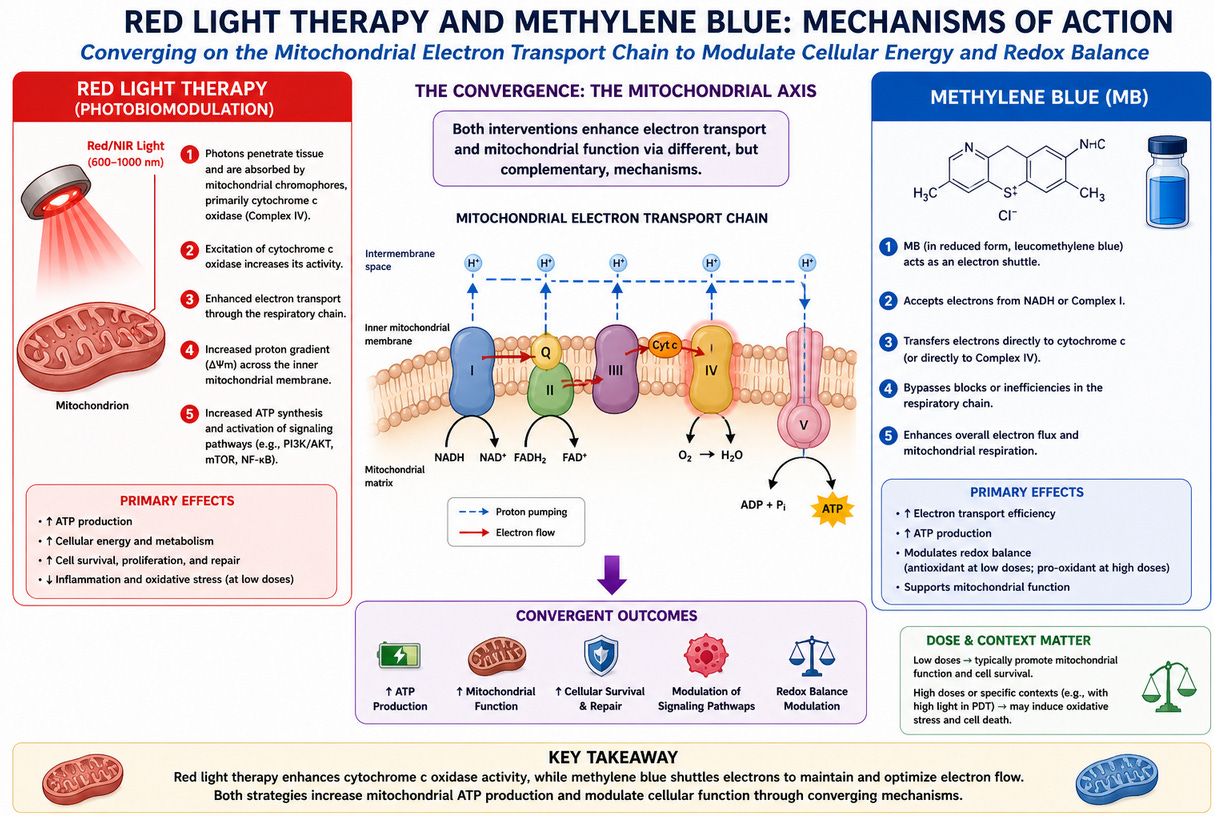
As outlined above, PBM works by stimulating cytochrome c oxidase, increasing:
ATP production
Mitochondrial activity
Cellular repair pathways
Pro-survival signaling (PI3K/AKT, mTOR)
In normal tissue, this is beneficial.
In cancer, it becomes complicated.
Because cancer cells—particularly cancer stem cells (CSCs)—are not metabolically “dead.” Many are highly adaptable and can shift toward oxidative phosphorylation (OXPHOS) when needed.
Now enter methylene blue.
Methylene Blue: Not Just a Dye — A Mitochondrial Drug
Methylene blue is often described casually, but biologically it is anything but trivial.
At low doses, it acts as:
An electron cycler within the electron transport chain
A bypass for dysfunctional mitochondria
A reducer of oxidative stress under certain conditions
A driver of mitochondrial efficiency and ATP production
In simple terms:
Methylene blue can make mitochondria work better.
Now combine that with red light.
The Combination: Amplifying the Same Axis
Red light therapy and methylene blue converge on the same biological target:
They both enhance mitochondrial respiration.
PBM → stimulates cytochrome c oxidase
Methylene blue → facilitates electron transfer
The net effect?
Increased oxidative phosphorylation, increased ATP, and enhanced cellular resilience
This is exactly why the combination is attractive in “longevity” circles.
It is also exactly why it raises concern in cancer.
This combination antagonizes the 5-axis metabolic approach to cancer, giving cancer the upper hand.
The Critical Question: Who Benefits More — The Patient or the Tumor?
This is the question that is almost never asked.
From a metabolic oncology perspective, many therapeutic strategies aim to:
Impair mitochondrial function
Increase metabolic stress
Reduce ATP availability
Target OXPHOS-dependent cancer stem cells
The PBM + methylene blue combination does the opposite:
Enhances mitochondrial efficiency
Increases ATP production
Supports cellular recovery under stress
So we are left with a fundamental tension:
Are we strengthening normal tissue—or rescuing metabolically stressed cancer cells?
The honest answer is:
We don’t know.
But the biology should give us pause.
“But Isn’t Methylene Blue Used to Kill Cancer Cells?”
Yes—and this is where confusion arises.
Methylene blue can act as a photosensitizer in photodynamic therapy (PDT).
Under specific conditions:
High local concentration
Targeted delivery
Controlled light wavelength and intensity
Adequate oxygen availability
…it can generate reactive oxygen species (ROS) that damage and kill tumor cells.
However, this is not what most people are doing.
There is a world of difference between:
1. Controlled oncologic photodynamic therapy
Defined drug dose
Tumor targeting
Specific light parameters
Clinician-guided
vs
2. Wellness-style red light + methylene blue
Variable dosing
Systemic exposure
Non-targeted light
No control of tumor microenvironment
These are not equivalent.
Not even close.
Dose and Context Change Everything
Methylene blue is a classic example of a biphasic agent:
Low dose → antioxidant, mitochondrial enhancer
High dose + light → pro-oxidant, cytotoxic
Red light therapy is also biphasic:
Low dose → stimulation and repair
Higher dose → inhibition or stress
So the combination is not inherently “good” or “bad.”
It is context-dependent.
But in cancer, context is everything.
The Tumor Microenvironment Matters
Cancer is not just a collection of cells—it is an ecosystem.
Within that ecosystem:
Hypoxia varies
Mitochondrial dependence varies
Immune surveillance varies
Redox balance is tightly regulated
Introducing:
A mitochondrial enhancer (methylene blue)
Plus a mitochondrial stimulator (PBM)
…may shift that ecosystem in unpredictable ways.
Particularly concerning are tumors that are:
Slow-growing
Metabolically flexible
OXPHOS-dependent
Examples include certain:
Neuroendocrine tumors
Hormone-driven cancers
Dormant or minimal residual disease states
In these settings, enhancing mitochondrial function may confer a survival advantage.
Clinical Reality: What Do We Actually Know?
Here is the honest, evidence-based position:
There is no strong clinical evidence that PBM + methylene blue accelerates cancer
There is also no robust evidence that it is safe in this context
Most data are:
Preclinical
Mechanistic
Highly context-dependent
Which brings us back to a fundamental principle of medicine:
Biological plausibility matters—especially when clinical data are lacking.
A Practical, Clinically Grounded Approach
Where this combination may be reasonable
Non-cancer patients
Neurologic or mitochondrial disorders (under supervision)
Carefully designed photodynamic therapy protocols
Where caution is warranted
Active cancer
Known primary tumors
Metastatic disease
Peri-treatment windows (chemo, radiation, metabolic therapies)
What I would avoid
Applying red/NIR light directly over tumors
Combining systemic methylene blue with local PBM over cancer sites
Whole-body PBM + methylene blue in active cancer
The Bottom Line
Methylene blue plus red light therapy is a powerful biological intervention.
But power cuts both ways.
At low doses, this combination may enhance mitochondrial function and cellular survival.
Under controlled conditions, it can be used to destroy cancer cells.
The difference is not subtle—it is dose, timing, targeting, and intent.
Until we have better data, the most rational approach is simple:
Do not casually apply a mitochondrial-enhancing therapy over a disease where mitochondrial control is central to survival.


I have just had a remarkable experience that i would like to share. I have had a constant stream of basal cell and squamus skin cancers for the last 30 years. My parents had 3 between them, total. I have had dozens frozen and cut off. I saw my dermatologist every 4 months.
Then my health plan changed, and I moved and didn’t go for a couple years. During that time I tried interesting ideas I found on the Internet. I have a red light sun lamp and take spirulina, which I understand is similar in effect to methylene blue.
I just went to the new dermatologist and he found nothing. No skin cancers whatsoever. I was floored. I am not sure if the results are attributable to this effort alone as I do other things as well, (DMSO and eggplant salve on anything that appears possibly precancerous, grape seed extract and vit C supplements).
I would like to find some doctor who finds this case medically interesting. To me it feels like I cured cancer. I would like to share this with anyone else who suffered through dozens of skin cancer surgeries like I did.
I think this is one of the most overlooked contradictions in metabolic cancer care.
So many patients are trying to metabolically stress cancer cells… while simultaneously adding therapies that may rescue mitochondrial function and ATP production.